- Number Of Protons And Electrons In Zinc
- Number Of Electrons In Outer Shell Zinc
- Number Of Unpaired Electrons In Zinc
- Number Of Unpaired Electrons In Zinc
- Number Of Electrons In Zinc
Zinc is a chemical element with atomic number 30 which means there are 30 protons and 30 electrons in the atomic structure. The chemical symbol for Zinc is Zn. Electron Configuration and Oxidation States of Zinc Electron configuration of Zinc is Ar 3d10 4s2. Calculate the number of moles of electrons transferred in the balanced equation, n. N = 4 moles of electrons. Substitute values into the Nernst equation and solve for the non-standard cell potential, E cell. E cell = + 0.152 V - (0.0257/4) ln(1.02 x 10 6) E cell = 0.063 V Top.
Electrochemical Cell Potentials
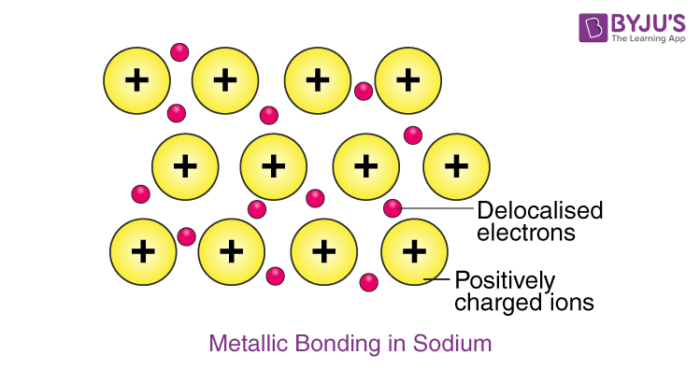
The precise definition involves quantum mechanics, but it is a number that characterizes the subshell. The third column is the maximum number of electrons that can be put into a subshell of that type. For example, the top row says that each s-type subshell (1s, 2s, etc.) can have at most two electrons in it. In each case the figure is 4 greater. A normal zinc (Zn) atom has 30 protons in its nuclear so that there are 30 electrons to make the atom be balanced of charge. The electrons are arranged by a rule that 1s2,2s2,2p6,3s2,3p6,4s2,3d10 (see more: electron configuration). Go to a periodic table and you'll see that the atomic number of zinc is 30. This means zinc has 30 electrons and 30 protons (its charge is null). The number of neutrons can vary.

The cell potential (voltage) for an electrochemical cell can be predicted from half-reactions and its operating conditions (chemical nature of materials, temperature, gas partial pressures, and concentrations).
Determining Standard State Cell PotentialsA cell's standard state potential is the potential of the cell under standard state conditions, which is approximated with concentrations of 1 mole per liter (1 M) and pressures of 1 atmosphere at 25oC.
Number Of Protons And Electrons In Zinc
To calculate the standard cell potential for a reaction
- Write the oxidation and reduction half-reactions for the cell.
- Look up the reduction potential, Eoreduction, for the reduction half-reaction in a table of reduction potentials
- Look up the reduction potential for the reverse of the oxidation half-reaction and reverse the sign to obtain the oxidation potential. For the oxidation half-reaction, Eooxidation = - Eoreduction.
- Add the potentials of the half-cells to get the overall standard cell potential.
Example: Find the standard cell potential for an electrochemical cell with the following cell reaction.

- Write the half-reactions for each process.
- Look up the standard potentials for the redcution half-reaction.
- Look up the standard reduction potential for the reverse of the oxidation reaction and change the sign.
- Add the cell potentials together to get the overall standard cell potential.
Cu2+(aq) + 2 e-' NOSAVE height=14 width=15> Cu(s)
Eooxidation of Zn = - ( - 0.762 V) = + 0.762 V
| oxidation: | Zn(s) ' NOSAVE height=14 width=15> Zn2+(aq) + 2 e- | Eoox. = - Eored. = - (- 0.762 V) = + 0.762 V |
| reduction: | Cu2+(aq) + 2 e-' NOSAVE height=14 width=15> Cu(s) | Eored. = + 0.339 V |
| overall: | Zn(s) + Cu2+(aq) ' NOSAVE height=14 width=15> Zn2+(aq) + Cu(s) | Eocell = + 1.101 V |
Determining Non-Standard State Cell Potentials
To determine the cell potential when the conditions are other than standard state (concentrations not 1 molar and/or pressures not 1 atmosphere):
- Determine the standard state cell potential.
- Determine the new cell potential resulting from the changed conditions.
- Determine Q, the reaction quotient.
- Deternine n, the number of electrons transferred in the reaction 'n'.
- Determine Ecell, the cell potential at the non-standard state conditions using the Nernst equation.
Ecell = cell potential at non-standard state conditions
Eocell = standard state cell potential
R = constant (8.31 J/mole K)
T = absolute temperature (Kelvin scale)
F = Faraday's constant (96,485 C/mole e-)
n = number of moles of electrons transferred in the balanced equation for the reaction occurring
in the cell
Q = reaction quotient for the reaction. aA + bB ' NOSAVE height=14 width=15> cC + dD,
Number Of Electrons In Outer Shell Zinc
If the temperature of the cell remains at 25oC, the equation simplifies to:
Ecell = Eocell - (0.0257/n) ln Q
or in terms of log10
Ecell = Eocell - (0.0592/n) log Q
Number Of Unpaired Electrons In Zinc
Example: Predict the cell potential for the following reaction when the pressure of the oxygen gas is 2.50 atm, the hydrogen ion concentration is 0.10 M, and the bromide ion concentration is 0.25 M.
Number Of Unpaired Electrons In Zinc
- Calculate the standard cell potential for the reaction, Eocell, using the tabled values:
- Determine the new cell potential resulting from the changed conditions.
- Calculate the value for the reaction quotient, Q. (Note: We calculate Q using molar concentrations for solutions and pressures for gases. Water and bromine are both liquids, therefore they are not included in the calculation of Q.)
- Calculate the number of moles of electrons transferred in the balanced equation, n.
- Substitute values into the Nernst equation and solve for the non-standard cell potential, Ecell.
| oxidation: | 4 Br-(aq) ' NOSAVE height=14 width=15> 2 Br2(l) + 4 e- | Eoox. = - Eored. = - (+ 1.077 V) = - 1.077 V |
| reduction: | O2(g) + 4 H+(aq) + 4 e- ' NOSAVE height=14 width=15> 2 H2O(l) | Eored. = + 1.229 V |
| overall: | O2(g) + 4 H+(aq) + 4 Br-(aq) ' NOSAVE height=14 width=15> 2 H2O(l) + 2 Br2(l) | Eocell = + 0.152 V |
Number Of Electrons In Zinc
Ecell = 0.063 V
